Toll Free Helpline (India): 1800 1234 070
Rest of World: +91-9810852116
Free Publication Certificate
Vol. 3, Issue 10 (2014)
Formulation and evaluation of controlled release colon targeted micro sponge of Aceclofenac
Author(s):
Shah Harsh, Karishma Patel, U.M.U padhyay
Shah Harsh, Karishma Patel, U.M.U padhyay
Abstract:
Aim - Continuous administration of therapeutic agent is desirable to maintain fixed plasma levels. Microsponges are porous, polymeric nanostructures that are mostly used for prolonged action.
Microsponges are designed to deliver a pharmaceutically active ingredient efficiently at minimum dose and also to enhance stability, reduce side effects, and modify drug release profiles. This system can work efficiently for systemic as well as local effect.
Experimental work - In present study, Aceclofenac microsponge formulation was prepared by Quasi emulsion solvent diffusion technique using Ethyl cellulose, Eudragit RS100, Eudragit S100 and Eudragit RL100 in different conc. Prepared microsponge were evaluated for % Practical yield, % Loading efficiency, particle size. Drug- excipients compatibility studies were performed by FTIR. Optimized batch of Aceclofenac microsponge was further formulated as tablet formulation for colon delivery. Prepared tablet formulations were evaluated for physical parameters like Pre & Post compression evaluation. The drug release data of optimized Batch were fitted into different kinetic models which show that the drug release from tablet formulations follows zero order release.
Result - Various batches were formulated using different drug polymer ratio and optimized batches were selected and further formulated as tablet and various parameters were being examined and batch MS 6 II was selected as the best batch which showed 97.55% of release in 24 hrs and studied for the various Kinetic models which show that the drug release from tablet formulations followed zero order release with r2 value of 0.99 and n value for korsmayer pepas shows n value 0.942 shows that it is non-fickian diffusion of drug release.
Conclusion - Aceclofenac was successfully encapsulated into Microsponge by Quasi emulsion solvent diffusion technique using Eudragit RS100 as polymer for enhancement of solubility, flow properties and compression characteristics and controlling the release rate upto 24 hrs. After stability study there were no physical changes and same drug release with Aceclofenac was observed. Hence, the batch MS 6 II was stable.
Aim - Continuous administration of therapeutic agent is desirable to maintain fixed plasma levels. Microsponges are porous, polymeric nanostructures that are mostly used for prolonged action.
Microsponges are designed to deliver a pharmaceutically active ingredient efficiently at minimum dose and also to enhance stability, reduce side effects, and modify drug release profiles. This system can work efficiently for systemic as well as local effect.
Experimental work - In present study, Aceclofenac microsponge formulation was prepared by Quasi emulsion solvent diffusion technique using Ethyl cellulose, Eudragit RS100, Eudragit S100 and Eudragit RL100 in different conc. Prepared microsponge were evaluated for % Practical yield, % Loading efficiency, particle size. Drug- excipients compatibility studies were performed by FTIR. Optimized batch of Aceclofenac microsponge was further formulated as tablet formulation for colon delivery. Prepared tablet formulations were evaluated for physical parameters like Pre & Post compression evaluation. The drug release data of optimized Batch were fitted into different kinetic models which show that the drug release from tablet formulations follows zero order release.
Result - Various batches were formulated using different drug polymer ratio and optimized batches were selected and further formulated as tablet and various parameters were being examined and batch MS 6 II was selected as the best batch which showed 97.55% of release in 24 hrs and studied for the various Kinetic models which show that the drug release from tablet formulations followed zero order release with r2 value of 0.99 and n value for korsmayer pepas shows n value 0.942 shows that it is non-fickian diffusion of drug release.
Conclusion - Aceclofenac was successfully encapsulated into Microsponge by Quasi emulsion solvent diffusion technique using Eudragit RS100 as polymer for enhancement of solubility, flow properties and compression characteristics and controlling the release rate upto 24 hrs. After stability study there were no physical changes and same drug release with Aceclofenac was observed. Hence, the batch MS 6 II was stable.
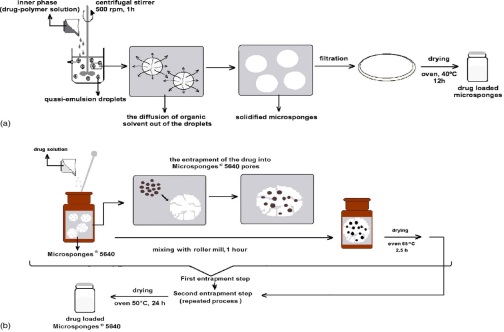
Fig.: Preparation of microsponges by quasi-emulsion solvent diffusion method
Pages: 81-87 | 2112 Views 240 Downloads

How to cite this article:
Shah Harsh, Karishma Patel, U.M.U padhyay. Formulation and evaluation of controlled release colon targeted micro sponge of Aceclofenac. Pharma Innovation 2014;3(10):81-87.







