Toll Free Helpline (India): 1800 1234 070
Rest of World: +91-9810852116
Free Publication Certificate
Vol. 3, Issue 4 (2014)
Formulation and in-vitro evaluation of oral disintegrating tablets containing solid dispersions of Candesartan Cilexetil
Author(s):
Ramya Krishna S
Ramya Krishna S
Abstract:
Candesartan Cilexetil is an angiotensin II receptor antagonist used mainly for the treatment of hypertension. The drug is having low solubility in biological fluids which results in poor bioavailability after oral administration. Hence present study was carried to enhance dissolution properties of candesartan cilexetil. Solid dispersions of candesartan cilexetil were prepared by using PEG-6000 as water soluble carries at various proportions 1:1, 1:2, 1:3. The kneading and solvent evaporation methods were used to prepare solid dispersions. The prepared dispersions were made into tablets by the direct compression method.
The release profile was studied in phosphate buffer pH 6.5 containing 0.35% polysorbate 20. It was found that the dissolution rate of tablets containing solid dispersions were higher than those of intact drug. The degree of dissolution rate enhancement depended on the amount of the carrier i.e., the higher the amount of the carrier used; the higher the dissolution rate was obtained. Among the prepared batches formulation F4 gave highest dissolution. The increase in the dissolution rate of the drug may be due to increase in wet ability, hydrophilic nature of the carrier and also possibility due to a reduction in drug crystalline.
Candesartan Cilexetil is an angiotensin II receptor antagonist used mainly for the treatment of hypertension. The drug is having low solubility in biological fluids which results in poor bioavailability after oral administration. Hence present study was carried to enhance dissolution properties of candesartan cilexetil. Solid dispersions of candesartan cilexetil were prepared by using PEG-6000 as water soluble carries at various proportions 1:1, 1:2, 1:3. The kneading and solvent evaporation methods were used to prepare solid dispersions. The prepared dispersions were made into tablets by the direct compression method.
The release profile was studied in phosphate buffer pH 6.5 containing 0.35% polysorbate 20. It was found that the dissolution rate of tablets containing solid dispersions were higher than those of intact drug. The degree of dissolution rate enhancement depended on the amount of the carrier i.e., the higher the amount of the carrier used; the higher the dissolution rate was obtained. Among the prepared batches formulation F4 gave highest dissolution. The increase in the dissolution rate of the drug may be due to increase in wet ability, hydrophilic nature of the carrier and also possibility due to a reduction in drug crystalline.
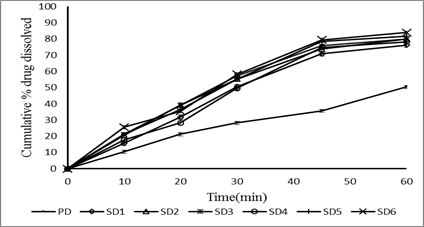
Fig.: comparative in vitro release profile of solid dispersions prepared by kneading method.
Pages: 72-78 | 1452 Views 81 Downloads

How to cite this article:
Ramya Krishna S. Formulation and in-vitro evaluation of oral disintegrating tablets containing solid dispersions of Candesartan Cilexetil. Pharma Innovation 2014;3(4):72-78.







