Toll Free Helpline (India): 1800 1234 070
Rest of World: +91-9810852116
Free Publication Certificate
Vol. 3, Issue 9 (2014)
Pathomorphological description of bronchial and lung arteries for patients with iii-iv stage of COPD under exacerbation
Author(s):
Malofiy L. S.
Malofiy L. S.
Abstract:
The paper shows the main morphological changes of the blood vessels of the lungs in COPD patients in the acute phase. It is established that the most evident changes occur in all the membranes of the arteries of the level of segmental and sub-segmental bronchi in the form of media hyperplasia, fibrinoid necrosis areas, dystrophic phenomena of vascular smooth muscle cell, structural transformation of internal and external elastic membranes, their hyperplastic, elastofibrosis, collapse and fragmentation, accretion of unsulfured glycosaminoglycans longwise fibers. Morphometrics show that media is 3-4 times thicker than intima and the lumen of the most arteries is narrowed down to 25-30% compared to the outer diameter. Moreover, at exacerbation of chronic obstructive pulmonary disease in III-IV stage there are observed lymphohistiocytic reaction longwise the arteries. Conducted comprehensive morphological studies indicate that not only the bronchial tree, but also bronchial arteries were involved in the pathological process, which leads to disorders of the different generation bronchi blood supply and disorders of normal functioning of the bronchi.
The paper shows the main morphological changes of the blood vessels of the lungs in COPD patients in the acute phase. It is established that the most evident changes occur in all the membranes of the arteries of the level of segmental and sub-segmental bronchi in the form of media hyperplasia, fibrinoid necrosis areas, dystrophic phenomena of vascular smooth muscle cell, structural transformation of internal and external elastic membranes, their hyperplastic, elastofibrosis, collapse and fragmentation, accretion of unsulfured glycosaminoglycans longwise fibers. Morphometrics show that media is 3-4 times thicker than intima and the lumen of the most arteries is narrowed down to 25-30% compared to the outer diameter. Moreover, at exacerbation of chronic obstructive pulmonary disease in III-IV stage there are observed lymphohistiocytic reaction longwise the arteries. Conducted comprehensive morphological studies indicate that not only the bronchial tree, but also bronchial arteries were involved in the pathological process, which leads to disorders of the different generation bronchi blood supply and disorders of normal functioning of the bronchi.
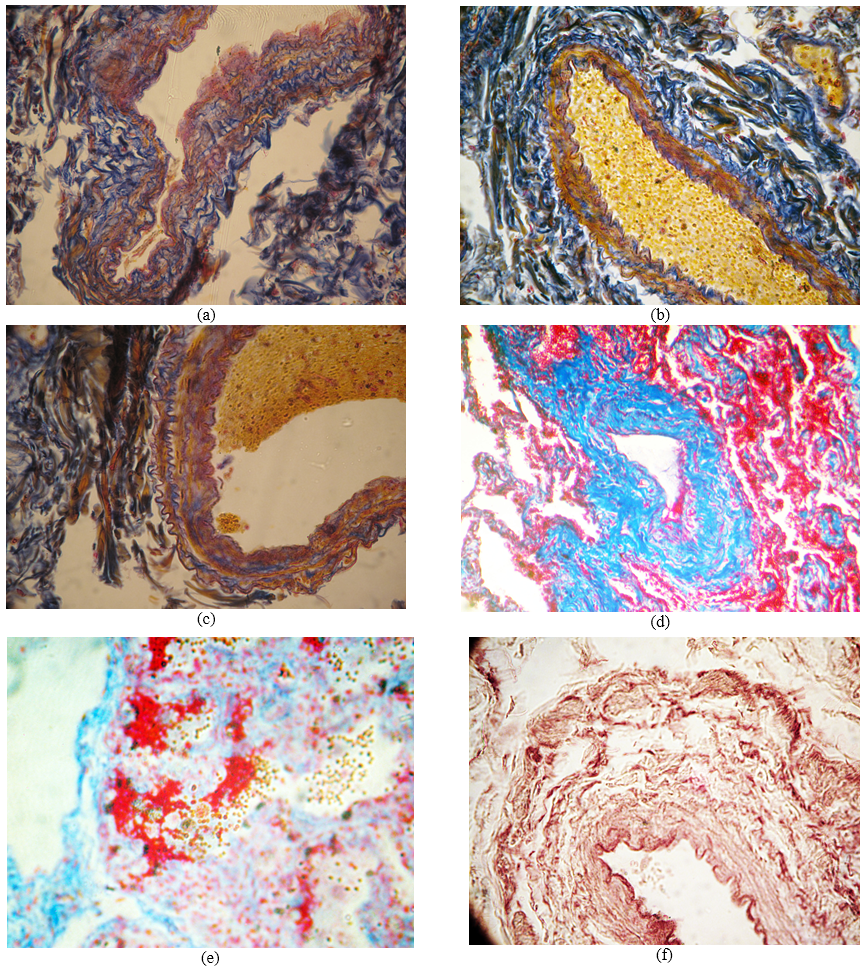
Fig.: The morphology of the bronchial arteries of the lungs in the acute phase of III-IV stage of COPD: a – bronchial artery wall with symptoms of disorganization; б - bronchial artery wall hyperelastosis; в – changes in luminal surface of epithelial layer and distinct bellows of external elastic membrane; г –hyperplasia of media; д – fibrinoid necrosis of the bronchial artery walls; e – lamination, fragmentation and disorganization of the bronchial artery elastic membranes. Staining: а-в – after Mason; г-д – by MSB method in Zerbino-Lukasevych modification; e – with fuchsin after Hart. Zooming: а-в – ocular 10, lens 10; г-е – ocular 10, lens 20.
Pages: 50-52 | 1573 Views 81 Downloads

How to cite this article:
Malofiy L. S.. Pathomorphological description of bronchial and lung arteries for patients with iii-iv stage of COPD under exacerbation. Pharma Innovation 2014;3(9):50-52.







