Toll Free Helpline (India): 1800 1234 070
Rest of World: +91-9810852116
Free Publication Certificate
Vol. 2, Issue 3 (2013)
Dabigatran Etexilate: A Drug Update
Author(s):
Kamalpreet Kaur, Dr. Vivek Gupta
Kamalpreet Kaur, Dr. Vivek Gupta
Abstract:
The Direct Thrombin Inhibitors are a new class of anticoagulants that binds directly to the thrombin enzyme and blocks its effect. DTI’s prevents the conversion of fibrinogen to fibrin by binding the activity of thrombin. Pradaxa is used to help prevent strokes or serious blood clots in patients with atrial fibrillation. Dabigatran has shown efficacy in the prevention of thromboembolism in Phase 2 trials in orthopedic surgery and atrial fibrillation. Dabigatran has also undergone extensive Phase 3 clinical trials for the prevention of primary thromboembolism. FDA advisory committee recommends approval of Dabigatran Etexilate for prevention of Stroke in Atrial Fibrillation. Dabigatran has been licensed for the Total Hip Replacement and Total Knee Replacement in over 75 countries, including Europe and Canada. Hence, this paper reviews the existing safety and efficacy data for the use of dabigatran etexilate and discusses the potential role of dabigatran in the management of VTE, THR, TKR, atrial fibrillation.
The Direct Thrombin Inhibitors are a new class of anticoagulants that binds directly to the thrombin enzyme and blocks its effect. DTI’s prevents the conversion of fibrinogen to fibrin by binding the activity of thrombin. Pradaxa is used to help prevent strokes or serious blood clots in patients with atrial fibrillation. Dabigatran has shown efficacy in the prevention of thromboembolism in Phase 2 trials in orthopedic surgery and atrial fibrillation. Dabigatran has also undergone extensive Phase 3 clinical trials for the prevention of primary thromboembolism. FDA advisory committee recommends approval of Dabigatran Etexilate for prevention of Stroke in Atrial Fibrillation. Dabigatran has been licensed for the Total Hip Replacement and Total Knee Replacement in over 75 countries, including Europe and Canada. Hence, this paper reviews the existing safety and efficacy data for the use of dabigatran etexilate and discusses the potential role of dabigatran in the management of VTE, THR, TKR, atrial fibrillation.
Related Graphics:
Click here for more related graphics
Click here for more related graphics
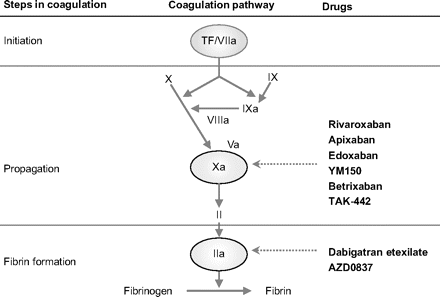
Fig.: fig 2
Pages: 141-148 | 1456 Views 82 Downloads

How to cite this article:
Kamalpreet Kaur, Dr. Vivek Gupta. Dabigatran Etexilate: A Drug Update. Pharma Innovation 2013;2(3):141-148.







